Pulse oximetry is a test used to measure the oxygen level (oxygen saturation) of the blood. It is an easy, painless measure of how well oxygen is being sent to parts of your body furthest from your heart, such as the arms and legs.
A clip-like device called a probe is placed on a body part, such as a finger or ear lobe. The probe uses light to measure how much oxygen is in the blood. This information helps the healthcare provider decide if a person needs extra oxygen.
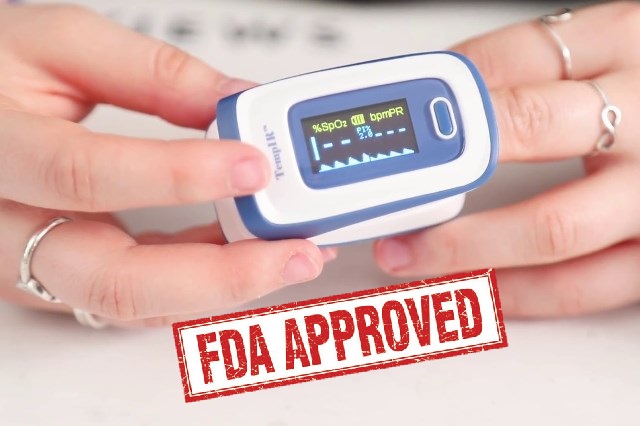
FDA Approved Pulse Oximeter list 2022
While you can purchase an at-home pulse oximeter, many of them are not necessarily medically-approved devices and are often designed for sports or recreational use.
Research has shown that the accuracy of at-home pulse oximeters cannot be validated. However, if you want to monitor your oxygen levels, these devices can be a good indicator of the state of your health if you start showing symptoms.
If you need a reliable pulse oximeter, look for one that is FDA approved, a few on the list below are.
AccuMed pulse oximeter
This finger pulse oximeter is designed for people who want to know their SpO2 (Blood Oxygen Saturation Levels) and pulse rate.
The compact and lightweight design allow you to take it with you and keep an eye on your health anytime, anywhere.
You can take measurements before or after any exercise, focusing on your own SpO2 and pulse changes, and more.
Wellue pulse oximeter
If you wonder, which pulse oximeters are FDA approved?
Medical devices, sold in the United States, such as pulse oximeters are regulated by the FDA. This is important because the FDA monitors and ensures that medical products that are only approved by them are sold on the market and are safe for use.
What are the benefits of these devices?
They show graphical reports of sleep and trends of blood oxygen level status and heart rate. The lightweight, small, and comfortable ring sensor for continuous overnight tracking is durable and fits most fingers.
Metene pulse oximeter
This product uses the most advanced photoelectric principle, through the high-quality transparent acrylic panel display.
Allows people to directly judge whether the measurement environment is suitable for measurement, to avoid the influence of strong light on the measurement results, so as to ensure high precision.
Innovo Fingertip pulse oximeter
This pulse oximeter features small volume, low power consumption, convenient operation, and is portable.
It is only necessary to place one of your fingers on a fingertip photoelectric sensor for diagnosis, and a screen will directly display the measured hemoglobin saturation value.
A pulse oximeter can be used to measure blood oxygen saturation (in percent of SpO2) and pulse rate. It should be used for spot monitoring and not for continuous monitoring.
Zacurate pulse oximeter
Now you can accurately and reliably determine your SpO2 (Blood Oxygen Saturation Levels), Pulse Rate, and Pulse Strength. Designed for sports enthusiasts, aviators, trainers, bikers, mountain climbers, or anyone who wants to obtain their SpO2 on the go.
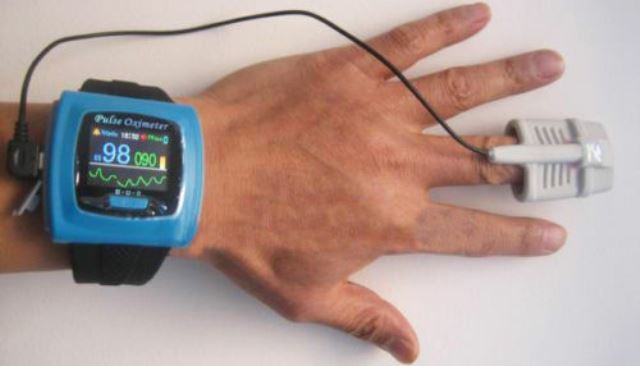
Wrist pulse oximeter
CONTEC Pulse Oximeter
The wrist pulse oximeter features a color, multi-direction OLED (organic light-emitting diode) screen with four levels of brightness.
The display shows pulse rate, blood oxygen saturation, pulse waveform, pulse bar graph, and battery level indicator.
- The pulse oximeter can sound an alarm in the following scenarios:
- when the pulse rate or oxygen saturation drops below a pre-set level
- when the oximeter unit isn’t correctly positioned on the finger
- when the battery level is low.
Accurately record pulse and SpO2 blood oxygen saturation. The comfortable design fits pediatric and adult users. Simple and easy-to-use wrist-worn design.
Pulse oximeter FDA cleared
We recommend you identify your device by the applicable regulation number and product code indicated and include the information described below.
It is recommended that you clarify whether the device and accessories are intended for (intended use):
- As a stand-alone device or a multi-parameter module.
- For use in spot-checking or continuous monitoring.
- For a single-use or multi-use.
- For out-of-hospital transport, or for home use.
It is recommended that you identify and describe:
- Scientific principles showing how the device achieves its intended use, i.e. the theory of operation.
- Recommended configuration, sensor geometry and application sites.
- Design features; for example, functions, alarms. In general, pulse oximeters intended for continuous monitoring should include pulse rate and SpO2 alarms.
- Specify all interface accessories; for example, patient cable, extension cables, sensors, bandages.
- If the device will be provided sterile; and if the device is a single-use reprocessed device.
FDA Approved medical devices
The FDA approved pulse oximeter list is a guideline for oximeters:
FDA’s guidance documents, including this guidance, do not establish legally enforceable responsibilities. Instead, guidances describe the Agency’s current thinking on a topic and should be viewed only as recommendations, unless specific regulatory or statutory requirements are cited.
The use of the word should in Agency guidance means that something is suggested or recommended, but not required.
- Pulse oximeter: An oximeter is a device used to transmit radiation at a known wavelength(s) through blood and to measure the blood oxygen saturation based on the amount of reflected or scattered radiation. It may be used alone or in conjunction with a fiberoptic oximeter catheter
- Ear oximeter: An ear oximeter is an extravascular device used to transmit light at a known wavelength(s) through blood in the ear. The amount of reflected or scattered light as indicated by this device is used to measure the blood oxygen saturation.
These classifications regulations group together all oximeters intended to measure blood oxygen saturation. The regulations include devices using reflectance, transmittance, and fiber optic technologies, which are collectively referred to as pulse oximeters for the purpose of this guidance.
The terms “transmittance” and “reflectance” refer to the sensor geometry and are not related to the principles of pulse oximetry and how the light is absorbed by hemoglobin.
How accurate are home pulse oximeters?
Pulse oximeters today are highly accurate. Medically graded versions for home use can deliver reliable results within 2% of what arterial blood gas will show. For those of you with severe oxygen issues, home devices may become less accurate if your readings fall below 80%, but you’ll be hospitalized in that event regardless of the equipment you have at home.
What is good oxygen saturation?
Normal blood oxygen ranges between 80 and 100 mm Hg. Normal readings from a pulse oximeter fall between 95 and 100. If you have a condition that affects your blood oxygen levels, such as COPD, your healthcare professional may have alternate ranges for your normal levels. Be sure to plan your readings with your healthcare professional, so you know what to look for.

Oxygen therapy at home
For patients on long‐term oxygen therapy, pulse oximetry arterial oxygen saturation (SaO2) measurements are unnecessary except to assess changes in clinical status or to facilitate changes in the oxygen prescription.
Home pulse oximetry is also indicated when there is a need to monitor the adequacy of SaO2 or the need to quantitate the response of SaO2 to a therapeutic intervention.
Overnight oximetry guidelines
Nocturnal home pulse oximetry has been used to assess nocturnal desaturation in patients with chronic obstructive pulmonary diseases (COPD). However, he found that nocturnal desaturation in COPD patients exhibited marked night-to-night variability when measured with home pulse oximetry.
A single home pulse oximetry reading at home may be insufficient for an accurate assessment of nocturnal desaturation. stated that for COPD patients with more profound diurnal hypercapnia, polysomnography is preferred over nocturnal pulse oximetry to rule out other coexisting sleep-related respiratory disorders.
Oximeters sold in the United States are FDA approved. This approval, officially recognized under a code called 510k certification.
The FDA provides several ways for you to check if the FDA approved or cleared a medical device or, as described below if the FDA authorized the device to be used during a public health emergency.
Check for Approved and Cleared Products in the Devices@FDA Database: Devices@FDA is a catalog of approved and cleared medical device information from the FDA. Go to the Devices@FDA Database.
Pulse Oximeters: Answering Your Frequently Asked Questions
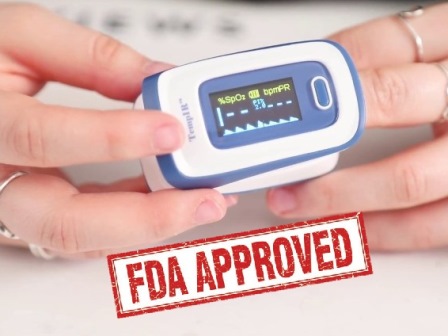
Which pulse oximeters are FDA Approved?
Oximeters sold in the United States are FDA approved. This approval is officially recognized under a code called 510k certification.
The most read

FDA approved Oximeter made in the USA
- Ever wonder if there are pulse oximeters made in the USA?
- How many medical device manufacturers are there in the United States?

Pulse Oximeters Price
- What is the best pulse oximeter?
- What is a normal oxygen saturation level?
- Are any oximeters made in the USA?
